|
Share this:
Read this article in PDF format
The laparoscopic cholecystectomy is considered the “gold standard” in chronic calculous cholecystitis treatment. After the gallbladder removal the physiology of gallbladder bile formation is changed.
Absence of the gallbladder leads to development of functional biliary hypertension and dilatation of common bile duct and the common hepatic duct (1-8). The dilatation of right and left hepatic ducts may be formed within 3-5 years after cholecystectomy (1-8). Functional hypertension in the common bile duct leads to development of functional hypertension in Wirsung's pancreatic duct accompanied by chronic pancreatitis symptoms (1-8).
During this period in some patients this is accompanied by chronic pancreatitis progression, dysfunction of the sphincter of Oddi and duodeno-gastral reflux (1-17). Duodeno-gastral reflux causes the development of atrophic (bile-acid-dependent) antral gastritis (1-11). After cholecystectomy 40% to 60% of patients suffer from dyspeptic disorders, 5% to 40% from pains of different localizations (1-17). Up to 70% of patients show symptoms of chronic “bland” intrahepatic cholestasis, chronic cholestatic hepatitis and compensatory bile-acid-dependent apoptosis of hepatocytes (11-15). In some of cholecystectomized patients with high concentration of hydrophobic hepatotoxic co-cancerogenic deoxycholic bile acid in serum and/or feces high risk of the colon cancer is found.
Therefore, depending on dysfunction (hyper tonus) or relaxation (hypo tonus) of the sphincter of Oddi, pathology in hepato-biliary-pancreato-duodenal-gastral zone will form after cholecystectomy.
Postcholecystectomy syndrome is a dysfunction of the sphincter of Oddi, caused by noncalculous obstructive disorder, which decrease bile passage and pancreatic juice outflow into the duodenum (1-2).
Diagnostic criteria of the sphincter Oddi dysfunction
- Complex of functional disorders continuing more than 3 months, among which the main clinic symptoms are recurrent severe and moderate pains lasting for more than 20 minutes, accompanied by nausea and/or vomiting and located in
- epigastrium or right hypochondrium with irradiation in the back and in the right scapular region (biliary type)
- left hypochondrium with irradiation in the back, abating when bending forward (pancreatic type)
- girdle pain (combined type)
- unstable stool with often cases of constipation or diarrhea
- dyspeptic syndrome
Pain may be accompanied by the following symptoms:
- pain occurs after meal
- pain occurs at night
- According to ultrasound examination data, dilatation of the common bile duct is up to 4-10 mm.
- The sphincter of Oddi incompetence is accompanied by increased hepatic bile passage into the duodenum and formation of duodeno-gastral reflux.
Causes of the biliary pain and chronic inflammation in the biliary tract and the sphincter of Oddi
- Pathology of the smooth muscle cells and epithelial cells in the biliary tract (high degree of COX-2 expression in the smooth muscle cells and epithelial cells of the bile ducts and the sphincter of Oddi).
- Hypersecretion of glycoprotein biliary mucin into bile ducts lumen (high degree of COX-2 expression in the epithelial cells of the bile ducts).
- Increased basal common bile duct resistance (high degree of COX-2 expression in the smooth muscle cells of the sphincter of Oddi).
Mechanism of development of pathologic disorders
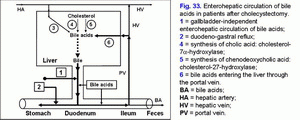
Absence of the gallbladder leads to surplus passage of hepatic bile only into the duodenum and increases frequency of gallbladder-independent enterohepatic circulation of bile acids. There is only gallbladder-independent enterohepatic circulation of bile acids in patients after cholecystectomy (fig. 33).
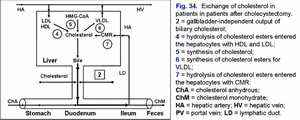
Absence of the gallbladder after cholecystectomy causes the increase in passage of hepatic bile into the duodenum and the gallbladder-independent enterohepatic circulation of biliary cholesterol and bilirubin (fig. 34).
Increase in gallbladder-independent enterohepatic circulation of bile acids causes increase in concentration of bile acids in the hepatocytes and decrease in the accumulation function and excretion function of the liver (i.e. formation of chronic “bland” intrahepatic cholestasis) (fig. 33).
Increase in the gallbladder-independent enterohepatic circulation of biliary cholesterol helps in increase of absorption of biliary cholesterol in the small intestine, the biliary cholesterol entering hepatocytes, and hypersecretion into hepatic bile (fig. 34).
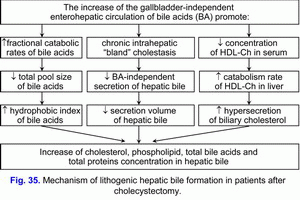
This factor contributes to the formation of the “lithogenic” hepatic bile and predisposes to choledocholithiasis (formation of gallstones in bile ducts) (fig. 35).
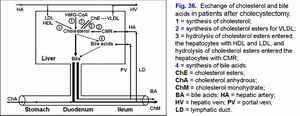
Increase in the gallbladder-dependent output of biliary cholesterol and in the concentration of total bile acids in duodenal bile cause causes the precipitation of cholesterol monohydrate crystals in the duodenum lumen in postcholecystectomic patients (fig. 36).
Due to the sphincter of Oddi incompetence surplus hepatic bile passage into the duodenum causes formation of duodeno-gastral reflux and development of chronic atrophic (bile-acid-dependent) antral gastritis, often accompanied by intestinal metaplasia, and gastroduodenitis (fig. 33, fig. 37).
Due to dysfunction of the sphincter of Oddi (high degree of COX-2 expression in the smooth muscle and epithelial cells of the sphincter of Oddi), hindered passage of hepatic bile into the duodenum causes development of the functional biliary hypertension, dilating of the common hepatic duct and common bile duct, development of the biliary pain in epigastrium or right hypochondrium (high degree of COX-2 expression in the smooth muscle and epithelial cells of the biliary tract), and chronic “bland” intrahepatic cholestasis and/or reactive hepatitis (fig. 38).
In some postcholecystectomic patients who have the sphincter of Oddi dysfunction (high degree of COX-2 expression in the smooth muscle and epithelial cells of the sphincter of Oddi), hindered passage of hepatic bile into the duodenum causes development of the functional biliary hypertension in the common bile duct, the functional hypertension in the Wirsung’s duct, the pain syndrome in the left hypochondrium, and the symptoms of chronic biliary pancreatitis (pancreatic type) (fig. 39).
Due to dysfunction of the sphincter of Oddi (high degree of COX-2 expression in the smooth muscle and epithelial cells of the sphincter of Oddi), hindered passage of hepatic bile into the duodenum causes development of the functional biliary hypertension dilating of the common hepatic duct and common bile duct, development of the biliary pain in epigastrium or right hypochondrium (high degree of COX-2 expression in the smooth muscle and epithelial cells of the biliary tract), chronic “bland” intrahepatic cholestasis and/or reactive hepatitis, the functional hypertension in the Wirsung’s duct, the pain syndrome in the left hypochondrium, and the symptoms of chronic biliary pancreatitis (combined type) (fig. 40).
Pathogenetic treatment of patients after cholecystectomy
Accordingly, treatment for patients after cholecystectomy (Postcholecystectomy syndrome is a dysfunction or incompetence of the sphincter of Oddi) (with biliary pain), aiming for the prophylactics of choledocholithiasis, duodeno-gastral reflux, antral atrophic (bile-acid-dependent) gastritis and chronic biliary pancreatitis includes
- Celecoxib - 100 mg, 2 times a day after meal for 5-7 days, after which
- Ursodeoxycholic acid - 750 mg, once a day in the evening for 2 month.
Celecoxib is a selective inhibitor of COX-2. Inhibiting COX-2 activity in the smooth muscle cells of the biliary tract and the sphincter of Oddi it brings relief of the biliary pain within 3-5 days, restoration of the passage of the hepatic bile into the duodenum.
Celecoxib is a selective inhibitor of COX-2, inhibiting COX-2 activity in the epithelial cells of the biliary tract mucosa causes decrease in secretion of glycoprotein mucin into the biliary tract lumen, concentration of the glycoprotein biliary mucin in the hepatic bile and viscosity of hepatic bile, which prevents formation of biliary sludge and gallstones in the common hepatic duct and common bile duct. Low COX-2 activity in the epithelial cells and the smooth muscle cells of the biliary tract helps in lowering the risk of choledocholithiasis development.
Ursodeoxycholic acid, is a hydrophilic hepatoprotective bile acid. It helps in dissolving the cholesterol monohydrate crystals in the biliary tract, decrease in lithogenicity of hepatic bile, disappearance of the chronic “bland” intrahepatic cholestasis (i.e. results in the restoration of the accumulation and excretion functions of liver), and in some patients helps in dissolving the biliary sludge in the biliary tract.
Ursodeoxycholic acid, is a hydrophilic hepatoprotective bile acid, decreasing aggressive properties of bile, prevents development of duodeno-gastral reflux and chronic atrophic (bile-acid-dependent) antral gastritis, often accompanied by intestinal metaplasia, and gastroduodenitis.
Celecoxib and Ursodeoxycholic acid, pathogenetically blocking main mechanisms of gallstone formation, help in prophylactics of gallstone formation in the biliary tract, and lower the risk of development of choledocholithiasis and chronic biliary pancreatitis.
Expected effectiveness is 90-95%.
Remission period is 18-24 months.
Attention!!! Information for patients:
Before using this scheme of treatment please check the contraindications (below) and side effects of using pharmacological preparations of Celecoxib and Ursodeoxycholic acid, and obtain your doctor’s permission.
Contraindications for Celecoxib:
- allergic reactions (nettle-rash, bronchial spasm) to acetylsalicylic acid or other NSAIDs (in anamnesis);
- 3rd trimester of pregnancy;
- high sensitivity to sulphonamides;
- high sensitivity to any component of the preparation.
Contraindications for Ursodeoxycholic acid:
- high sensitivity to the preparation;
- acute inflammatory diseases of the gallbladder and the bile ducts;
- ulcerative colitis;
- Crone’s disease.
This web page does not bear any legal responsibility for the use of the proposed treatment schemes without consulting your doctor.
References:
- Extrahepatic biliary system diseases: the gallbladder dysfunction and states after cholecystectomy. International Bulletin: gastroenterology. 2001; 6: 1-4.
- Ilchenko A.A. Postcholecystectomy syndrome: clinical aspects of problem. Consilium Medicum. Gastroenterology. 2006; 2.
- Komarov F.I., Galkin V.A., Ivanov A.I., Maksimov V.A. Associated diseases of organs of duodenocholedochopancreatic zone. M.: Medicine, 1983: 1-256.
- Zubovski G.A., ed. Radio and ultrasonic diagnosis of biliary tract diseases. Moscow: Medicine, 1987: 1-240.
- Barthet M., Affriat C., Bernard J.P. et al. Is biliary lithiasis associated with pancreatographic changes? Gut. 1995; 36(5): 761-765.
- Barthet M., Spinoza S., Affriat C. et al. Influence of age and biliary lithiasis on the diameter of the common bile duct. Gastroenterol. clin. Biol. 1995; 19: 156-160.
- Potter G.D. Bile acid diarrhea. Dig. Dis. Sci. 1998; 16: 118-124.
- Popovic O., Mivolic V., Kostic K. et al. Bile acid-mediated postcholecystectomy diarrhea // Gastroenterol. Inter. 1988; 1: Abstr. 790.
- Portincasa P., van de Meeberg P., van Erpecum K.J. et al. An update on the pathogenesis and treatment of cholesterol gallstones. Scand. J. Gastroenterol. 1997; 223: 60-69.
- Portincasa P., di Ciaula A., Palmieri V. et al. Impaired gallbladder and gastric motility and pathological gastroesophagal reflux in gallstone patients. Europ. J. clin. Invest. 1997; 27: 653-661.
- Fort J.M., Azpiroz F., Casellas F. et al. Bowel habit after cholecystectomy: physiological changes and clinical implications. Gastroenterology. 1996; 111: 617-622.
- Isogai M., Yamaguchi A., Hori A., Nakano S. Hepatic histopathological changes in biliary pancreatitis. Amer. J. Gastroenterol. 1995; 90(3): 449-454.
- Mulvihill S.J. Surgical management of gallstone disease and postoperative complications. In: M. Feldman, B.F. Scharschmidt, M.H. Sleisenger, eds. Sleisenger and Fordtran’s Gastrointestinal and Liver Disease: Pathophysiology, Diagnosis, Management. 6th ed. Philadelphia: WB Saunders Company, 1998: 973-984.
- Honda A., Yoshida T., Tanaka N., Matsuzaki Y., He B., Shoda J., Osuga T. Increased bile acid concentration in liver tissue with cholesterol gallstone disease. J Gastroenterol 1995; 30(1): 61-66.
- Geraghty J.M., Goldin R.D. Liver changes associated with cholecystitis. J Clin Pathol 1994; 47(5): 457-60.
- Carey M.C., Duane W.C. Enterohepatic circulation. In: Arias I.M., Boyer J.L., Fausto N., Jakoby W.B., Schachter D.A., Shafritz D.A., eds. The Liver, Biology and Pathobiology. 3rd ed. New York: Raven Press, 1994: 719-767.
- Hofmann A.F. Bile secretion and the enterohepatic circulation of bile acids. In: Feldman M., Scharschmidt B.F., Sleisenger M.H., eds. Sleisenger and Fordtran’s Gastrointestinal and Liver Disease: Pathophysiology, Diagnosis, Management. 6th ed. Philadelphia: WB Saunders Company, 1998: 937-948.
References (Celecoxib and UDCA):
- Chen XW, Cai JT. The impact of selective cycloxygenase-2 inhibitor celexibo on the formation of cholesterol gallstone. Zhonghua Nei Ke Za Zhi. 2003; 42(11): 797-9.
- Guarino MP, Carotti S, Sarzano M, Alloni R, Vanni M, Grosso M, Sironi G, Maffettone PL, Cicala M. Short-term ursodeoxycholic acid treatment improves gallbladder bile turnover in gallstone patients: a randomized trial. Neurogastroenterol Motil. 2005; 17(5): 680-6.
- Pazzi P, Petroni ML, Prandini N, Adam JA, Gullini S, Northfield TC, Jazrawi RP. Postprandial refilling and turnover: specific gallbladder motor function defects in patients with gallstone recurrence. Eur J Gastroenterol Hepatol. 2000; 12(7): 787-94.
- Ikegami T, Matsuzaki Y, Fukushima S, Shoda J, Olivier JL, Bouscarel B, Tanaka N. Suppressive effect of ursodeoxycholic acid on type IIA phospholipase A2 expression in HepG2 cells. Hepatology. 2005; 41(4): 896-905.
- Shoda J, Ueda T, Kawamoto T, Todoroki T, Asano T, Sugimoto Y, Ichikawa A, Maruyama T, Nimura Y, Tanaka N. Prostaglandin E receptors in bile ducts of hepatolithiasis patients and the pathobiological significance for cholangitis. Clin Gastroenterol Hepatol. 2003; 1(4): 285-96.
- Shoda J, Kano M, Asano T, Irimura T, Ueda T, Iwasaki R, Furukawa M, Kamiya J, Nimura Y, Todoroki T, Matsuzaki Y, Tanaka N. Secretory low-molecular-weight phospholipases A2 and their specific receptor in bile ducts of patients with intrahepatic calculi: factors of chronic proliferative cholangitis. Hepatology. 1999; 29(4): 1026-36.
- Tomida S, Abei M, Yamaguchi T, Matsuzaki Y, Shoda J, Tanaka N, Osuga T. Long-term ursodeoxycholic acid therapy is associated with reduced risk of biliary pain and acute cholecystitis in patients with gallbladder stones: a cohort analysis. Hepatology. 1999; 30(1): 6-13.
- Kano M, Shoda J, Irimura T, Ueda T, Iwasaki R, Urasaki T, Kawauchi Y, Asano T, Matsuzaki Y, Tanaka N. Effects of long-term ursodeoxycholate administration on expression levels of secretory low-molecular-weight phospholipases A2 and mucin genes in gallbladders and biliary composition in patients with multiple cholesterol stones. Hepatology. 1998; 28(2): 302-13.
- Shoda J, Ueda T, Ikegami T, Matsuzaki Y, Satoh S, Kano M, Matsuura K, Tanaka N. Increased biliary group II phospholipase A2 and altered gallbladder bile in patients with multiple cholesterol stones. Gastroenterology. 1997; 112(6): 2036-47.
- Carotti S, Guarino MP, Cicala M, Perrone G, Alloni R, Segreto F, Rabitti C, Morini S. Effect of ursodeoxycholic acid on inflammatory infiltrate in gallbladder muscle of cholesterol gallstone patients. Neurogastroenterol Motil. 2010.
- Guarino MP, Carotti S, Morini S, Perrone G, Behar J, Altomare A, Alloni R, Caviglia R, Emerenziani S, Rabitti C, Cicala M. Decreased number of activated macrophages in gallbladder muscle layer of cholesterol gallstone patients following ursodeoxycholic acid. Gut. 2008; 57(12): 1740-1.
- Jüngst C, Sreejayan N, Zündt B, Müller I, Spelsberg FW, Hüttl TP, Kullak-Ublick GA, del Pozo R, Jüngst D, von Ritter C. Ursodeoxycholic acid reduces lipid peroxidation and mucin secretagogue activity in gallbladder bile of patients with cholesterol gallstones. Eur J Clin Invest. 2008; 38(9): 634-9.
- Spier BJ, Pfau PR, Lorenze KR, Knechtle SJ, Said A. Risk factors and outcomes in post-liver transplantation bile duct stones and casts: A case-control study. Liver Transpl. 2008; 14(10): 1461-5.
- Guarino MP, Cong P, Cicala M, Alloni R, Carotti S, Behar J. Ursodeoxycholic acid improves muscle contractility and inflammation in symptomatic gallbladders with cholesterol gallstones. Gut. 2007; 56(6): 815-20.
- Mas MR, Comert B, Mas N, Yamanel L, Ozotuk H, Tasci I, Jazrawi RP. Effects of long term hydrophilic bile acid therapy on in vitro contraction of gallbladder muscle strips in patients with cholesterol gallstones. World J Gastroenterol. 2007; 13(32): 4336-9.
- Jüngst C, Sreejayan N, Eder MI, von Stillfried N, Zündt B, Spelsberg FW, Kullak-Ublick GA, Jüngst D, von Ritter C. Lipid peroxidation and mucin secretagogue activity in bile of gallstone patients. Eur J Clin Invest. 2007; 37(9): 731-6.
- Itoh S, Kono M, Akimoto T. Psoriasis treated with ursodeoxycholic acid: three case reports. Clin Exp Dermatol. 2007; 32(4): 398-400.
- Beuers U. Drug insight: Mechanisms and sites of action of ursodeoxycholic acid in cholestasis. Nat Clin Pract Gastroenterol Hepatol. 2006; 3(6): 318-28.
- Colecchia A, Mazzella G, Sandri L, Azzaroli F, Magliuolo M, Simoni P, Bacchi-Reggiani ML, Roda E, Festi D. Ursodeoxycholic acid improves gastrointestinal motility defects in gallstone patients. World J Gastroenterol. 2006; 12(33): 5336-43.
- Pemberton PW, Aboutwerat A, Smith A, Warnes TW. Ursodeoxycholic acid in primary biliary cirrhosis improves glutathione status but fails to reduce lipid peroxidation. Redox Rep. 2006; 11(3): 117-23.
- Jeong HJ, Kim CG. Pretreatment with ursodeoxycholic acid (UDCA) as a novel pharmacological intervention in hepatobiliary scintigraphy. Yonsei Med J. 2005; 46(3): 394-8.
- Fischer S, Müller I, Zündt BZ, Jüngst C, Meyer G, Jüngst D. Ursodeoxycholic acid decreases viscosity and sedimentable fractions of gallbladder bile in patients with cholesterol gallstones. Eur J Gastroenterol Hepatol. 2004; 16(3): 305-11.
- Sauter GH, Thiessen K, Parhofer KG, Jüngst C, Fischer S, Jüngst D. Effects of ursodeoxycholic acid on synthesis of cholesterol and bile acids in healthy subjects. Digestion. 2004; 70(2): 79-83.
- Xiao ZL, Biancani P, Carey MC, Behar J. Hydrophilic but not hydrophobic bile acids prevent gallbladder muscle dysfunction in acute cholecystitis. Hepatology. 2003; 37(6): 1442-50.
- Tazuma S, Nishioka T, Ochi H, Hyogo H, Sunami Y, Nakai K, Tsuboi K, Asamoto Y, Sakomoto M, Numata Y, Kanno K, Yamaguchi A, Kobuke T, Komichi D, Nonaka Y, Chayama K. Impaired gallbladder mucosal function in aged gallstone patients suppresses gallstone recurrence after successful extracorporeal shockwave lithotripsy. J Gastroenterol Hepatol. 2003; 18(2): 157-61.
- Gunsar C, Melek M, Karaca I, Sencan A, Mir E, Ortac R, Canan O. The biochemical and histopathological effects of ursodeoxycholic acid and metronidazole on total parenteral nutrition-associated hepatic dysfunction: an experimental study. Hepatogastroenterology. 2002; 49(44): 497-500.
- Xiao ZL, Rho AK, Biancani P, Behar J. Effects of bile acids on the muscle functions of guinea pig gallbladder. Am J Physiol Gastrointest Liver Physiol. 2002; 283(1): G87-94.
- Kano M, Shoda J, Satoh S, Kobayashi M, Matsuzaki Y, Abei M, Tanaka N. Increased expression of gallbladder cholecystokinin: a receptor in prairie dogs fed a high-cholesterol diet and its dissociation with decreased contractility in response to cholecystokinin. J Lab Clin Med. 2002; 139(5): 285-94.
- Wang DQ, Tazuma S. Effect of beta-muricholic acid on the prevention and dissolution of cholesterol gallstones in C57L/J mice. J Lipid Res. 2002; 43(11): 1960-8.
- Lukivskaya OY, Maskevich AA, Buko VU. Effect of ursodeoxycholic acid on prostaglandin metabolism and microsomal membranes in alcoholic fatty liver. Alcohol. 2001; 25(2): 99-105.
- Bomzon A, Ljubuncic P. Ursodeoxycholic acid and in vitro vasoactivity of hydrophobic bile acids. Dig Dis Sci. 2001; 46(9): 2017-24.
- Sunami Y, Tazuma S, Kajiyama G. Gallbladder dysfunction enhances physical density but not biochemical metastability of biliary vesicles. Dig Dis Sci. 2000; 45(12): 2382-91.
- Ljubuncic P, Said O, Ehrlich Y, Meddings JB, Shaffer EA, Bomzon A. On the in vitro vasoactivity of bile acids. Br J Pharmacol. 2000; 131(3): 387-98.
- Nishioka T, Tazuma S, Yamashita G, Kajiyama G. Partial replacement of bile salts causes marked changes of cholesterol crystallization in supersaturated model bile systems. Biochem J. 1999; 340 ( Pt 2): 445-51.
- Sinisalo J, Vanhanen H, Pajunen P, Vapaatalo H, Nieminen MS. Ursodeoxycholic acid and endothelial-dependent, nitric oxide-independent vasodilatation of forearm resistance arteries in patients with coronary heart disease. Br J Clin Pharmacol. 1999; 47(6): 661-5.
- van de Heijning BJ, van de Meeberg PC, Portincasa P, Doornewaard H, Hoebers FJ, van Erpecum KJ, Vanberge-Henegouwen GP. Effects of ursodeoxycholic acid therapy on in vitro gallbladder contractility in patients with cholesterol gallstones. Dig Dis Sci. 1999; 44(1): 190-6.
- Mendez-Sanchez N, Brink MA, Paigen B, Carey MC. Ursodeoxycholic acid and cholesterol induce enterohepatic cycling of bilirubin in rodents. Gastroenterology. 1998; 115(3): 722-32.
- Benedetti A, Alvaro D, Bassotti C, Gigliozzi A, Ferretti G, La Rosa T, Di Sario A, Baiocchi L, Jezequel AM. Cytotoxicity of bile salts against biliary epithelium: a study in isolated bile ductule fragments and isolated perfused rat liver. Hepatology. 1997; 26(1): 9-21.
- Ohtake M, Sandoh N, Sakaguchi T, Tsukada K, Hatakeyama K. Enhancement of portal blood flow by ursodesoxycholic acid in partially hepatectomized rats. Surg Today. 1996; 26(2): 142-4.
- Fahey DA, Carey MC, Donovan JM. Bile acid/phosphatidylcholine interactions in mixed monomolecular layers: differences in condensation effects but not interfacial orientation between hydrophobic and hydrophilic bile acid species. Biochemistry. 1995; 34(34): 10886-97.
- Bouscarel B, Ceryak S, Robins SJ, Fromm H. Studies on the mechanism of the ursodeoxycholic acid-induced increase in hepatic low-density lipoprotein binding. Lipids. 1995; 30(7): 607-17.
- Bomzon A, Ljubuncic P. Bile acids as endogenous vasodilators? Biochem Pharmacol. 1995; 49(5): 581-9.
- Jazrawi RP, Pazzi P, Petroni ML, Prandini N, Paul C, Adam JA, Gullini S, Northfield TC. Postprandial gallbladder motor function: refilling and turnover of bile in health and in cholelithiasis. Gastroenterology. 1995; 109(2): 582-91.
- Pak JM, Adeagbo AS, Triggle CR, Shaffer EA, Lee SS. Mechanism of bile salt vasoactivity: dependence on calcium channels in vascular smooth muscle. Br J Pharmacol. 1994; 112(4): 1209-15.
- Sasaki H, Tazuma S, Kajiyama G. Effects of 16,16-dimethyl prostaglandin E2 on biliary mucous glycoprotein and gallstone formation in guinea pigs. Scand J Gastroenterol. 1993; 28(6): 495-9.
- Mizuno S, Tazuma S, Kajiyama G. Stabilization of biliary lipid particles by ursodeoxycholic acid. Prolonged nucleation time in human gallbladder bile. Dig Dis Sci. 1993; 38(4): 684-93.
- Pak JM, Lee SS. Vasoactive effects of bile salts in cirrhotic rats: in vivo and in vitro studies. Hepatology. 1993; 18(5): 1175-81.
- Das JB, Cosentino CM, Levy MF, Ansari GG, Raffensperger JG. Early hepatobiliary dysfunction during total parenteral nutrition: an experimental study. J Pediatr Surg. 1993; 28(1): 14-8.
- Fromm H, Malavolti M. Bile acid dissolution therapy of gallbladder stones. Baillieres Clin Gastroenterol. 1992; 6(4): 689-95.
- Tazuma S, Sasaki H, Mizuno S, Sagawa H, Hashiba S, Horiuchi I, Kajiyama G. Effect of ursodeoxycholic acid administration on nucleation time in human gallbladder bile. Gastroenterology. 1989; 97(1): 173-8.
|